views

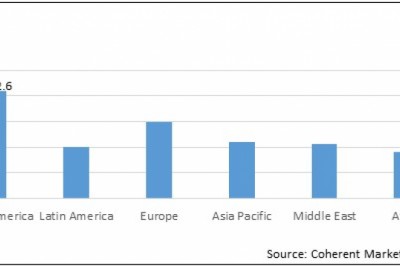
Since the start of March 2020, 976 organizations have reported disruptions to clinical trials due to COVID-19 (1), while as of July, 3,985 new clinical trials have launched in search of a vaccine or treatment for the disease (2).
The rapid growth of clinical trials for COVID-19
Trials by numbers
Since the start of March 2020, 976 organizations have reported disruptions to clinical trials due to COVID-19 (1), while as of July, 3,985 new clinical trials have launched in search of a vaccine or treatment for the disease (2).
Trial areas experiencing delays include Oncology, with 31.2% of trials delayed as of May 2020, followed by Central Nervous System at 17.5%, Gastrointestinal at 8.5%, Infectious Disease at 8.1%, and Cardiovascular at 8.1% (4). The most significant impact of the pandemic on pre-COVID-19 trials has been on patient recruitment (5), with 69.9% of trials suspending enrollment. Slow enrollment and the delayed initiation of planned trials have had the next largest impact at 17.3% and 12.8% respectively, with the lack of sites and investigators playing a key role in these delays (4).