views
In Vitro Diagnostics Industry Overview
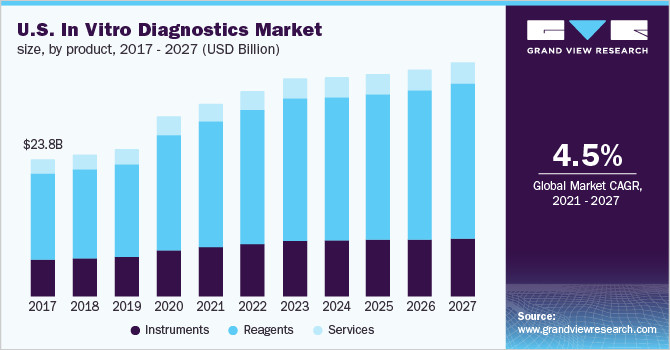
The global in vitro diagnostics market size was valued at USD 83.4 billion in 2020 and is expected to expand at a compound annual growth rate (CAGR) of 4.5% from 2021 to 2027.
The growth can be attributed to the increasing adoption of in vitro diagnostics (IVD) owing to a rise in testing due to the pandemic. The development of automated in vitro diagnostic systems for laboratories and hospitals to provide efficient, accurate, and error-free diagnosis is expected to fuel market growth. The rising number of in vitro diagnostic products being launched by key players is also fueling the lucrative growth rate of the market.
Globally, the elderly population is growing at a faster rate than the younger. In many countries, the growth of the older population can be attributed to declining fertility and low birth rate. According to United Nations, it was estimated that, in 2017, there were around 962 million people aged above 60 years worldwide.
Gather more insights about the market drivers, restrains and growth of the Global In Vitro Diagnostics Market
In addition, the number of individuals aged 80 years and above is projected to increase by threefold from 137 million in 2017 to 425 million in 2050. With age, the immune system is affected, which increases susceptibility to acquiring various diseases. Hence, a large geriatric population requires better healthcare, especially for chronic diseases.
The COVID-19 outbreak, caused due to SARS-CoV-2, is affecting millions of people globally. The outbreak of the pandemic has impelled the diagnostics industry into action, with a race to develop novel and rapid diagnostics kits for the detection of coronavirus. A majority of the tests are approved under Emergency Use Authorization (EUA) by federal agencies. Major assays used for COVID-19 detection employ the RT-PCR technique. Although the conventional test takes 5 to 6 hours for result output, PoC assays have reduced the duration significantly.
The prevalence of various diseases such as cancer, autoimmune diseases, and inflammatory conditions is increasing worldwide and is expected to drive the demand for in vitro diagnostic testing. For instance, according to the American Cancer Society, in 2018, about 1.7 million new cancer cases were reported and around 609,640 individuals died due to cancer.
About Grand View Research
Grand View Research, U.S.-based market research and consulting company, provides syndicated as well as customized research reports and consulting services. Registered in California and headquartered in San Francisco, the company comprises over 425 analysts and consultants, adding more than 1200 market research reports to its vast database each year. These reports offer in-depth analysis on 46 industries across 25 major countries worldwide. With the help of an interactive market intelligence platform, Grand View Research Helps Fortune 500 companies and renowned academic institutes understand the global and regional business environment and gauge the opportunities that lie ahead.h